Louanne Hudgins, M.D.
Changes in water level may disperse or concentrate populations and change the availability of food and water and access to potential toxins symptoms of pneumonia purchase generic tolterodine online. Estimation of whether biting insect populations have increased can be important symptoms 2 year molars generic tolterodine 4mg otc, as they may serve as disease vectors when administering medications 001mg is equal to order tolterodine no prescription. Water quality may be important as poor water quality may contribute to disease and mortality symptoms 3 days before period buy tolterodine 4 mg. Primary contamination by toxic substances can also lead to morbidity and mortality. Information on the condition and behaviour of animals prior to the outbreak should be recorded if possible, as should any changes in their abundance and distribution. Specific features of problem areas Other specific features not mentioned above should be noted and provided to the diagnostician. Supplementary investigations If further investigations are carried out these reports should be summarised and kept as a supplement to the original findings. These reports should be copied to the diagnostic laboratory where the specimens were sent. Chapter 1, Field manual of wildlife diseases: general field procedures and diseases of birds. Deciding whether a disease should be managed or not, rests largely on the extent to which it endangers human and animal health and welfare, economic systems, conservation aspirations, and the likelihood that intervention will achieve disease management objectives. The appropriate approach will depend on the characteristics of the problem and, when dealing with an infectious disease, on the correct identification of reservoirs, hosts and vectors of infection. Prevention and control of a disease is usually more easily achieved than complete eradication [>Section 3. Appropriate disease management options will depend on whether one is dealing with endemic or epidemic disease, and whether the intention is to prevent or control disease spread. Disposal options are varied and again need to be used with caution in wetland situations to reduce risks of pollution of water courses or further spread of infection. Targeting vectors in integrated disease control strategies can be effective and usually take the form of environmental management, biological controls and/or chemical controls, or actions to reduce the contact between susceptible hosts and vectors. Vaccination programmes, often supplemented by other disease control measures, can help control and even eliminate diseases affecting livestock. Habitat modification in wetlands can eliminate or reduce the risk of disease, by reducing the prevalence of disease-causing agents, vectors and/or hosts and their contact with one another, through the manipulation of wetland hydrology, vegetation and topography and alterations in host distribution and density. Complete eradication of a disease requires a thorough understanding of its epidemiology, sufficient political and stakeholder support and thorough resourcing and is thus rarely achieved! Sanitation measures involve preventing animal contact with physical, microbiological, biological or chemical agents of disease, which are often found in wastes, and maintaining clean, hygienic conditions. Inadequate sanitation is a major cause of disease worldwide and simple measures for improving sanitation are known to have significant beneficial impacts on public and animal health. Disinfection prevents the mechanical transmission of disease agents from one location to another by animals and inanimate objects, by eliminating many or all pathogenic microorganisms (except bacterial spores) on inanimate objects so that they will no longer serve as a source of infection. Disinfection following fieldwork prevents transfer of infection on fomites such as boots and clothing. Measures taken to prevent a disease outbreak For public health and biosecurity reasons, people working in wetlands should maintain high standards of sanitation and hygiene, and avoid direct contact with human and animal faeces, solid wastes, domestic, industrial and agricultural wastes [>Section 3. Effective sanitation and hygiene can be achieved through engineering solutions. Livestock housing should be regularly cleaned and disinfected and waste and clean water should be separated and safely stored. Waste materials from captive animals should be properly processed and disposed of. Cleaning is a necessary first step that allows the subsequent disinfecting agent to come into direct contact with pathogens on the surfaces of an object. Some viruses, bacteria and other infectious agents can persist in the environment for protracted periods. Disinfection is only practical for circumstances in which the pathogen or disease transmission occurs in a very limited area. The appropriateness of disinfectants will be informed by information on the presence of non-target species and other potential environmental impacts, particularly any adverse effects on wetland ecosystem function. Disinfection for wildlife disease situations is often difficult and likely to be most effective where wild animals are concentrated, such as at artificial feeding or watering sites. Measures taken during a disease outbreak During a disease outbreak, it may be necessary (if practical) to disinfect the local environment to prevent recurrence. Procedures are generally similar, however, the nature and infectivity of the pathogen will affect the protocols employed. For example, chytrid fungus and foot and mouth disease virus will require very different procedures for decontamination. As a consequence, disinfection of a disease outbreak site should always be conducted under the guidance of disease control specialists. From the above, the following should be done, as appropriate: during disinfection activities, easily cleaned protective clothes such as waterproof coveralls and rubber boots and gloves should be worn, and all clothes should be thoroughly washed after use and before leaving the outbreak area. If possible, personnel should wash their hair before leaving the area, and always before going to other wetland areas. Personnel handling potentially infectious agents should not work with similar species or those susceptible to disease for at least seven days after participating in disease control activities. Disinfection processes require a suitable disinfectant, containers for the solution once it has been diluted to the appropriate strength and a suitable method for its application. Vehicles and boats with pumps and tanks can be used to store and dispense disinfectant. All vehicles should be cleaned and disinfected on entering and leaving an outbreak area. Brushes, buckets, and containers that can be used to clean and disinfect boots and pressure sprayers that can be used to dispense the disinfectant are also required. Disease control specialists should advise on the most appropriate type of disinfectant and its application in wetland settings. Physical and chemical factors: temperature, pH, relative humidity, and water hardness. Organic and inorganic matter: serum, blood, pus, faeces or other organic materials can interfere with the effectiveness of disinfectants. Duration of exposure: items must be exposed to the chemical for the appropriate contact time. Disease control contingency plans should identify readily available sources of supplies and equipment needed for disinfection activities in case of an outbreak. Wetland managers, particularly those caring for housed livestock, should consider keeping a supply of disinfectant for general use. Health and safety risks of using chemicals Disinfectants may be toxic to humans as well as animals and plants, and therefore all chemicals should be used in accordance with the relevant safety precautions. Key factors that help to assess the human health risk of chemical exposure include the duration, intensity. Wetland managers may be responsible for informing workers about the chemical hazards involved and implementing disinfection control measures. Where required, wetland managers should be able to readily provide workers with appropriate personal protective equipment and Material Safety Data Sheets (usually available on the internet) for each chemical or mixture of chemicals that may be in use. Animal health authorities should be contacted to advise on appropriate measures remembering that the health and safety of the personnel involved in any disposal operation are paramount. Rapid and effectively planned carcase collection and disposal is essential to prevent spread of infectious disease and to reduce potential secondary poisoning in the case of toxic diseases. Presented below is a broad overview of the most commonly used methods for animal carcase collection and disposal, each has strengths and weaknesses which should be considered in the context of each specific situation. Collection of carcases Ideally carcases can be dealt with in situ to reduce chances of spread of infectious agents. However, in most circumstances where an outbreak has occurred and there are a number of carcases, they will need to be gathered to a central location for disposal. To help prevent potentially contaminated body fluids leaking during collection and transport to the central location, wherever possible (depending on size of dead animal), the carcases should be double bagged in plastic leak-proof bags (noting that claws, beaks etc. Wooden containers are difficult to decontaminate as fluids soak into wood so, wherever possible, plastic or metal bins/barrows etc. If carcases are being transported off-site to disposal facilities this must be done in leak-proof vehicles. Advice should be sought from animal health authorities regarding transportation of potentially infectious carcasses. Burial of carcases this is the often a preferred method of disposal as it is relatively easy to organise, quick, inexpensive, has potentially fewer immediate environmental hazards and it is a convenient means of disposing of large numbers of carcases.
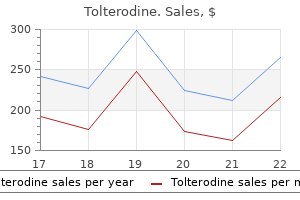
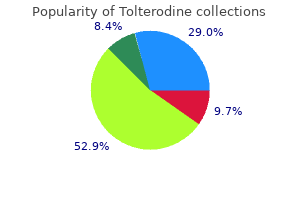
To 30 identify these studies medicine 3d printing cheap 2mg tolterodine with mastercard, a PubMed search (articles 20 published before September 2012) was carried out with the strategy ((nocturia medications hair loss order tolterodine 4mg mastercard. Population-based (including community-based) studies 0 0 20 30 40 50 60 70 80 published in English and conducted among both Age (years) sexes with a wide age range (at least 40 years) which reported percentage data were included treatment action campaign tolterodine 1mg with amex. This effect persisted medicine 5852 buy tolterodine online pills, although attenuated (39,40), after adjusting for comorbidities and socio-economic status. In secondary care?seeking populations (45,46), black women also reported more nocturia. Nocturia 143 In small studies in Taiwan (48,49) and Scotland (50), associations between nocturia and ethnicity were found. Indeed, in most studies, many of the potentially causal factors of nocturia, such as sleep apnea, remained unmeasured. Furthermore, in the recent Internetbased study, response rates were highest among whites and lowest among blacks (42). Earlier research has shown that lower response rates are related with higher prevalence estimates (51,52). Overall, the underlying mechanisms for the possible association of nocturia with race/ethnicity remain unknown. This is probably due to several reasons, including: 1) longitudinal studies are more difficult to perform than cross-sectional studies; and 2) there is uncertainty about the definition of incident nocturia (53?55) and about the appropriate time interval for repeated sampling (56). The fluctuation of nocturia contributes to these uncertainties (57), and as research into nocturia is relatively young, it is unsurprising that only few longitudinal studies exist (11). Incidence of nocturia was defined by following those with no nocturia at baseline (defined as 0 or 1 episodes) to year 2. Younger cohorts (50 years old) had a lower incidence (61 cases/1,000 person-years) than did the two older cohorts (60and 70-year cohorts, 91 and 93 cases/1,000 person-years, respectively). In all age cohorts, the observed incidence was higher during the second 5-year period than during the first: 102, 168, and 167 cases per 1,000 person-years, respectively. However, one should interpret this somewhat cautiously, as the prevalence for 60-year-olds was higher at 10-year follow-up than was the prevalence for 70-year-olds at baseline. The incidence of nocturia at 3 or more times per night (from 2 or fewer times/night) was substantially less across all age groups (3, 12, and 16 cases/1,000 personyears, respectively, for the age cohorts). This study is difficult to compare with other studies, as no information on incidence of nocturia from <1 episode to >2 or more episodes is available (59). Follow-up was performed 11 years later when 3,257 men replied (3,000 men had died, and 691 had emigrated or were not available in the register). The authors presented prevalence data at two time points: at least 2 voids per night was reported by 13% at baseline and by 50% after 11 years. However, the authors concluded (57) that due to this fluctuation it is almost impossible to provide reliable incidence rates of nocturia in community-dwelling older men. In Denmark, 278 (of 305) women responded at 5 years and 242 at 12 years after their first pregnancy (62,63). Of those 69 women who had nocturia during pregnancy, it had resolved in 99% of women (68/69) at 3 months postpartum (62). Sleep disturbance is an important mechanism underlying subjective complaints and broader health consequences of nocturia. This observation was consistent across all age groups and increased with age, from 39. Data from the 2003 National Sleep Foundation Survey in America corroborates these findings among older adults aged 55?84 years, with more than half of the respondents reporting nocturia as the cause of disturbed sleep, 4-fold higher than pain?the next most common reason cited for disturbed sleep (65). However, it is also possible that other conditions, such as insomnia or depression, can lead to sleep disruption, and nocturnal voids once awake then represent a secondary phenomenon (14,15). Sleep loss associated with nocturia has been associated with daytime fatigue and reduced quality of life (69). Bother associated with nocturia has been shown to be related to both quantity and quality of sleep (70?72). Difficulty initiating sleep, as well as difficulty returning to sleep after an awakening play an important role (73,74). Nocturia 145 the frequency of nightly voids that constitutes a threshold for significant or meaningful impact on well-being is a pertinent practical and theoretical question. Although more frequent nocturia causes more bother, the relationship is not perfectly correlated (19,28,29,32,69,75?79). Similarly, two voiding episodes per night were associated with substantial impact on health-related quality of life compared with those without nocturia, and at least three episodes of nocturia resulted in further impairment of similar magnitude (19). These results indicate that two episodes of nocturia constitute a threshold beyond which nocturia has adverse effects on well-being, whereas 1 void per night does not identify subjects with interference from nocturia (19). An association of nocturia with depression has also been consistent across studies (80?82). However, untreated depression at baseline was predictive of incident nocturia (83). How much reduction in nocturia is needed to be clinically important has not been answered. As nocturia is associated with other factors that may affect bother and quality of life (comorbid conditions, lifestyle factors), not all bother is explained by number of voiding episodes. Hence, treatment for nocturia episodes may not relieve all impairment among subjects with nocturia (19). Sleep loss alters carbohydrate metabolism and endocrine function, and has been associated with incident diabetes (87,88). Additionally, falls constitute the greatest risk factor for fractures among the elderly (89). Nocturia is associated with an increased risk for both falls and fractures (90?96). Increased risk for mortality has been reported not only in the elderly, but also among younger men and women (33,93,97,98). In a Japanese study among the elderly, at least 2 voids per night was associated with a doubled risk for fractures and mortality (96). Data from the Krimpen study among older men (55?84 years of age) showed no association of nocturia with increased mortality risk after accounting for confounding factors (99). In contrast, results of the Olmsted County Study in men 60 years and older showed an almost 50% increase in mortality risk after multivariate analyses (100). Increased risk for morbidity and mortality among younger age groups and among those without prevalent comorbid conditions may indicate nocturia as a marker for impending morbidity. Although no research articles have been published on economic impact of nocturia, a few studies have investigated the impact of nocturia and associated sleep loss on work productivity. A study of 203 working adults in Sweden has shown reduced work productivity with nocturia. The prevalence of nocturia is higher among young women than young men, but the prevalence increases more markedly with age in men. Incidence of nocturia increases with age, but signifcant shortterm fuctuation in nocturia severity in individuals makes studies on incidence challenging. Two or more episodes of nocturia per night constitutes clinically meaningful nocturia severity in the general population, affecting quality of life and perceived health, while a single episode usually does not. Nocturia has been suggested to increase risk for falls, fractures, death, and impaired productivity. Approaches to identify intra-individual variation and other confounding infuences need to be considered. Longitudinal studies using high-quality methodology remain a priority requirement. Nocturia of twice or more per night may be a threshold of clinical signifcance in the general population. However, this threshold is not irrefutably established, and should not be extracted to sub-populations. Research into all grades of nocturia severity may yield information of clinical relevance. What has changed is the knowledge that nocturia has multi-factorial etiology, which is not always urological in origin. In normal adult urinary physiology, the amount of urine made at night is less than the functional bladder capacity during the daytime, and hence adults void more during the daytime and can sleep at night without having to wake up to void. Therefore, urine production and storage at night is based on two simple physiological events: the first is that the person needs to go to sleep and the second is that urine needs to be produced and stored in the bladder. If, for whatever reason, one or both of these two mechanisms are disturbed, then nocturia will result. 1mg tolterodine fast delivery. Is Your Dizziness Vertigo (BPPV)? Answer 4 Questions to Know.
Botulinum toxin is a metabolic product of the Gram-positive treatment laryngitis generic tolterodine 4 mg with visa, sporeforming bacterium Clostridium botulinum medicine man dr dre order generic tolterodine canada. The toxin in high doses can cause the disease known as botulism medications requiring aims testing purchase discount tolterodine on line, a type of severe poisoning often acquired by consuming food that has become spoiled and contaminated with botulinum bacteria medications containing sulfa buy tolterodine on line. The latency period to the onset of symptoms ranges from 4 to 6 hours, but may be up to 14 days in extreme cases. After an initial bout of gastroenteritis, followed by central nerSchematic illustration of the biologically active botulinum toxin protein vous disturbances such as light fickering before the eyes, double vicomplex. Apart from its use in the treatment of various other cholinergic synapses, where it inhibits release of the neuro1 neurological disorders, botulinum toxin has become established as transmitter acetylcholine, leading to muscular paralysis of the affecthe predominant treatment in aesthetic medicine. Internalization Botulinum toxin is a two-chain polypeptide consisting of a light chain 3. The fnal active form of the protein is a complex make-up of plasma membrane of the cholinergic nerve endings. This binding to the two-chain neurotoxin itself, together with hemagglutinins and the presynaptic membrane shows a high degree of affnity and spenon-toxin, non-hemagglutinin proteins. The heavy H-chain of the toxin allows the large been decoded and show a high degree of homology to one another. The H-chain separates from the L-chain as the disulfde originating from a Clostridium species, which is why these substanbond is broken. This allows the L-chain to enter the cytoplasm of the ces are also jointly referred to as clostridial neurotoxins. The botulinum toxin light chain acts as a zinc-dependent endopeptiType A botulinum toxin is the main serotype in therapeutic use, espedase with proteolytic activity. In the cytosol, depending on serotype, it cially with regard to aesthetic indications. The depolarization triggered by this leads to contraction of the muscle 1 fbers. They form a complex with the synaptobrevin, which is integrated into the vesicle membrane and anchors it to the internal neuron membrane. According to the product insert, once reconstituted, the solutistopper should be cleaned with 70% alcohol before inserting the on should be used within hours. The NaCl solution is the authors have taken all reasonable care in preparing drawn in directly by the negative pressure in the vial. They do not assume any liability or guarantee for the updated the vial is now carefully swirled until the substance has dissolved fulstatus, accuracy and completeness of the product inforly in the saline. The ready-to-use solution can now be Should the latest information or further explanation drawn up into suitable syringes (cf. Figures do also apply to other product names containing identical substance preparations after similar reconstitution process. The given information also applies to other product names containing identical substance preparations. Preparing a two-third dilution the two-third dilution is used by the senior author when he apdo this, 0. This gives with a maximal diffusion range of the active substance are indicated 4 units per 0. Two-level injection of the basic rules is that the injections have to be as painless as pos-. Directed injection well as a careful insertion technique, are elementary in that context. Pain can be caused when the needle is inserted too deeply so that it pushes against the muscle-underlying periost and gets bent. To preWhich technique is recommended in individual cases is dependent vent this, needles always should be inserted slowly and diagonally to on the target muscle and the individual anatomical and functional the skin surface. A general distinction is made between deep injections, which administer the substance directly into the muscle belly and Video: Basic rules of injection superfcial injections applying the substance in a subcutaneous level. The elbow of the injection arm should ideally be mical slaps, s/he creates mechanical defection. The syringe is held betso immediately before the injection is done, the practiween the index and middle fngers, with the thumb placed loosely tioner is able to decrease the injection pain by up to on the plunger. The injection hand is supported on the outer edge of 80% as clinical experience has shown. Two fngers of the non-injecting hand can be used to fx and lightly compress the target muscle for a more precise injecting. The needle should be inserted gently and in a diagonal direction to the skin surface. Besides prevention of pain, there are further basic rules important to by the epicranial aponeurosis (Galea aponeurotica), which functions follow during the injecting in order to avoid possible unwanted side as a natural diffusion barrier. Along with careless injections, it is poseffects of the therapy with botulinum toxin A. As an example, supersible that the epicranial aponeurosis gets damaged by the needle alfcial subcutaneous injections or the use of dilutions. For that reason, it is important to inject with a maximal disareas with a given risk of overcorrection like the forehead. Ptosis can occur after unwanted distributions along the eyebrow without the given risk of ptosis. Overcorrection, by overly weakening the muscle, can intenbe given a suffcient distance from the eyebrows. This can often be remedied by soft tissue augmentation in the eyebrow produces a cosmetically undesirable, sad or tired or angry facial exregion and forehead. Advanced injectors typically treat these areas at the when injecting into the frontalis muscle and, in particular, into its same time. Superfcial injections at low dosages using the subdermal wheal technique are most appropriate in this respect. In some cases, it is worth doing the treatment in two sessions: the Caution: the lower part of the frontalis muscle (up to a distance of glabella is treated frst and the result of this awaited, as the procerus approx. Horizontal lines on the forehead Treatment Injection 5 Activation Injection technique the practitioner instructs the patient to contract the muscle actively: Raise Direct injection with vertical insertion of the needle in the medial region. Products and doses Complications / Managing complications Injection site Product Units/point Ml solution/point Medial frontalis Xeomin 2 0. If necessary, the ptosis may be remedied by soft tissue augmentation in the eyebrow Table 5. If the central parts of the frontalis muscle are treated, its lateral Higher doses may be needed in patients with a very high forehead. A separts may allow a certain amount of contraction in the outer recond treatment line may need to be placed above the frst if this is the case. When performing this correction, care must be taken to Age / inactive facial expressions: Factor 0. Combined treatment options the lower 2 cm are often left alone when treating the frontalis It may be a good idea to supplement the treatment with superfcial muscle. Small, augmentation of residual lines or with supportive skin regeneration comma-shaped lines may form above the brows. These fne lines therapy by percutaneous collagen induction, alias Medical Needling. Caution There is considerable variability in the anatomy of the forehead as well as the strength of the forehead musculature, which means there is always a risk of an overtreatment and resultant eyebrow ptosis. It Video: Treatment of horizontal forehead lines is essential for the injector to try to avoid this. In this context, it will be necessary to evaluate to what tal lines in the glabellar region by partial or complete deactivation of extent any residual function should be preserved. Repeated treatment of the glabellar region at 4-month internially into the central parts of the muscle vals over a period of 1 to 2 years encourages smoothing of these Corrugator supercilii muscle: one injection medially, directed along lines, thus leading to a lasting effect accompanied by a reduction in the fbers (other lateral injection points can also be considered) the chronically hypertrophic muscle segments. Orbicularis oculi muscle, orbital part: one superfcial injection (further injections along the eyebrow may be attached since a brow lift is often wished at the same time). Glabella (frown lines) Treatment Injection 5 Activation Injection technique the practitioner instructs the patient to contract the muscle actively: Pull Procerus muscle: direct injection with vertical needle insertion your eyebrows together towards the nose and down, or Frown. Products and doses Complications / Managing complications Injection site Product Units/point Ml solution/point Procerus Xeomin 2 0. Even if suffcient relaxation of the relevant muscles has been achieved, some individuals can produce frown lines voluntarily by recruiting the medial parts of the orbicularis oculi muscle. If this Correction factor is the case, small superfcial doses can be given into the palpable Man with active expressions: Factor 2 areas of activity of the orbicularis oculi muscle.
If pre-auto bone marrow is positive for disease involvement treatment xdr tb 1mg tolterodine visa, then pre-allo bone marrow should be bilateral medicine net generic 2 mg tolterodine with amex. See Tables 10 and 11 for post-transplant evaluations and additional lab instructions medications zovirax order tolterodine mastercard. Post-Autograft Evaluation Post-autografting workup is to be performed according to institutional Standard Practice guidelines medicine vial caps order 1 mg tolterodine. If 90 days passed after the auto-graft, the patient should undergo all evaluation guidelines detailed under the Pre-Allograft Evaluation (next section). Pre-Allograft Evaluation the routine pre-allografting workup is to be performed according to institutional Standard Practice and is to include the following: i. Careful physical exam with determination of Karnofsky score (Appendix A) or Lansky Play score (Appendix B) and findings related to underlying malignancy. Additionally, see above tables 8 and 9 for disease specific pre-transplant evaluations. Post-allograft Evaluation See Table 10 for disease specific post-transplant evaluation on Day +28, 56, 84, etc. Electrolyte panel, renal and hepatic function three times a week until day 28 and then once per week until tacrolimus is stopped, unless clinical circumstances suggest the need for more frequent evaluations. First-line therapy will be reinstitution of tacrolimus, if previously discontinued, and administration of high-dose corticosteroids with subsequent taper as described in the Standard Practice Guidelines. Every effort will be made to determine the exact cause of death for all patients as they occur. See text and additional table for pre-transplant evaluation and lab information Disease Specimen/ Test/ Imaging Clinical/ Comment Days Years Annual x Research 28 56 84 180 1 1. All instructions apply to both preand post-transplant evaluations unless identified otherwise. Refer to Standard Practice of the institution for information regarding administration, toxicity and complications. Description: Fludarabine monophosphate is a purine antimetabolite that, after administration, undergoes rapid conversion in plasma to the nucleoside 2-fluoro ara-A (F-araA). Storage and Administration: Fludarabine monophosphate is commercially available as a 50 mg/vial which is reconstituted with 2 ml of sterile water, resulting in a 25mg/ml solution. Side Effects and Toxicity: Clinical toxicities of fludarabine monophosphate include: myelosuppression, primarily lymphopenia and granulocytopenia, alopecia, rash, dermatitis, nausea, vomiting, anorexia, stomatitis, diarrhea, somnolence, fatigue, peripheral neuropathy, mental status changes, cortical blindness, hepatocellular toxicity with elevation in serum transaminases, and interstitial pneumonitis. Immunosuppression observed with the use of fludarabine increases the risk of infection which can be life-threatening C. Storage and Administration: Cyclophosphamide for injection is commercially available in 2000 mg vials which are reconstituted with 100 ml sterile water for injection. The calculated dose will be diluted further in 250-500 ml of Dextrose 5% in water. Description: Mesna is a prophylactic agent used to prevent hemorrhagic cystitis induced by the oxazaphosphorines (cyclophosphamide and ifosfamide). Mesna binds with acrolein, the neurotoxic metabolite produced by the oxazaphosphorines, to produce a non-toxic thioether and slows the rate of acrolein formation by combining with 4-hydroxy metabolites of oxazaphosphorines. Storage and Administration: Mesna is commercially available in 200 mg, 400 mg and 1000 mg vials containing a 100 mg/ml solution. Each dose of mesna will be diluted further in 50 ml of normal saline to be infused over 15 min. The total daily dose of mesna is equal to 80% of the total daily dose of cyclophosphamide. Side Effects and Toxicity: At the doses used for uroprotection mesna is virtually non-toxic. However, adverse effects which may be attributable to mesna include nausea and vomiting, diarrhea, abdominal pain, altered taste, rash, urticaria, headache, joint or limb pain, hypotension and fatigue. Calcineurin mediates the first intracellular signal required for T-cell activation after antigen recognition by the T-cell receptor. It is also used for immunosuppression after kidney, cardiac, pancreas, pancreatic islet cell and small bowel transplantation. It is metabolized in the liver by unknown mechanisms, but demethylation and hydroxylation have been proposed based on in vitro studies. Storage and Administration: Tacrolimus is commercially available in capsule form (0. Drugs that may increase blood levels of tacrolimus include: macrolide antibiotics, antifungals (fluconazole and itraconazole), calcium channel blockers, cimetidine, danazol, methylprednisolone and metoclopramide. Drugs that may decrease blood levels of tacrolimus include: phenobarbital, phenytoin, carbamazepine, rifamycins and the anti-fungal agent caspofungin. As a result of the inhibition of de novo purine synthesis, proliferation of Tand B-lymphocytes is blocked and antibody production is inhibited. There are no pharmacokinetic interactions with ganciclovir, cotrimoxazole, oral contraceptives or cyclosporine. The oral formulation is supplied in 250mg hard gelatin capsules and can be stored at room temperature. Side effects and toxicity: Side effect profiles include diarrhea, leukopenia, sepsis, allergic reactions, and vomiting. In the setting of marrow transplantation, several etiologic factors may contribute to alterations in gastrointestinal and hematologic parameters. Most patients in these studies were also on other drugs known to be associated with these complications. With the exception of hypophosphatemia there seems to be no difference in side effects between i. Flow of information concerning clinical trial participants originates with the clinicians and nurses in the clinic or referring clinicians at other institutions and is transmitted to the trial coordinator. Thus, multiple health care providers provide independent observations and participate in monitoring this trial. If determination of adverse events is controversial, coinvestigators will convene on an ad hoc basis as necessary to review the primary data and render a decision. These guidelines detail the expedited reporting requirements, definitions of particular events. Furthermore, an additional safeguard for adverse event analysis and reporting in this protocol is provided by stopping rules and interim analysis. These are reviewed for eligibility, adherence to the protocol, accuracy, and completeness by the study staff. The investigator will ensure that data collected conform to all established guidelines for coding collection, key entry and verification. Any publication or presentation will refer to patients by this number and not by name. The licensed medical records department, affiliated with the institution where the patient receives medical care, maintains all original inpatient and outpatient chart documents. Access is restricted to personnel authorized by the Division of Clinical Research. Ongoing data collection will be monitored by the coordinating center to ensure data completeness and uniformity across study centers. The case report form provides detailed descriptions of the transplantation procedure, disease response, and complications. Referring physicians are required to report the date and cause of death within 48 hours of occurrence. Supporting medical records must be available upon request to substantiate data on the case report forms. Death information including date and cause will be reviewed from these medical records, and will be confirmed by death certificate. In case of a cross-over from this protocol to another tandem autologous-allogeneic research protocol (#1409 or other appropriate protocol) or vice versa, enrollment will be counted on the crossed-over protocol if the patient receives the allogeneic transplantation on that protocol. If the patient is crossed-over but does not receive the allogeneic transplantation, enrollment will be counted on the initial protocol. Reconstitution of lymphocyte subsets in peripheral blood at is a secondary endpoint. Patients may continue to be enrolled pending evaluation of the safety endpoints in each cohort of 10 patients; however, the outcome of subsequent patients cannot be used to override a stopping rule triggered in the earlier cohort. The operating characteristics of these stopping rules are summarized in the table below. In addition to the above outcomes, secondary endpoints to be examined include overall survival, relapse rate, and engraftment. |